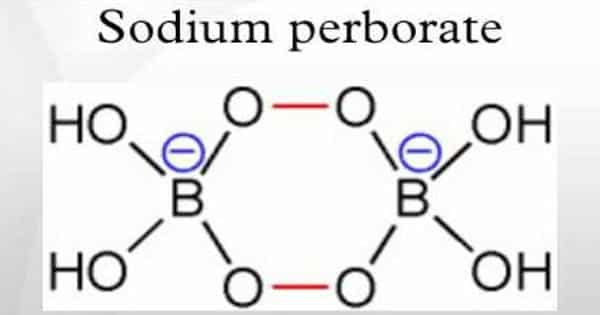
Sodium perborate monohydrate is commonly used as an oxidizing agent and disinfectant. While it may have some sterilizing properties, it is typically not recommended as the sole method for sterilizing surgical instruments, including laparoscopic instruments. However, it can be used as a cleaning agent in combination with other sterilization methods. Here’s a general procedure for using sodium perborate monohydrate to clean laparoscopic instruments:
1. Pre-cleaning: Start by removing any visible debris or organic matter from the instruments. Rinse them with water to remove initial contaminants.
2. Preparation: Prepare a cleaning solution by dissolving sodium perborate monohydrate in water according to the manufacturer’s instructions. The concentration and contact time may vary, so refer to the product label for specific guidelines.
3. Immersion: Submerge the laparoscopic instruments in the prepared cleaning solution, ensuring they are fully covered. Allow them to soak for the recommended duration. This typically ranges from a few minutes to several hours, depending on the concentration and instructions provided by the manufacturer.
4. Agitation: During the soaking period, gently agitate the instruments to facilitate the removal of any remaining debris or contaminants. This can be done by gently swirling the container or using a soft brush to scrub hard-to-reach areas.
5. Rinse: After the recommended contact time, remove the instruments from the cleaning solution and thoroughly rinse them with clean water. Ensure that all residues from the cleaning solution are completely removed.
6. Drying and further sterilization: Once the instruments are rinsed, dry them with a clean, lint-free cloth or allow them to air dry. Keep in mind that sterilization is a separate process, and sodium perborate monohydrate alone may not provide sufficient sterilization. Depending on the specific requirements, you may need to use additional sterilization methods such as autoclaving, ethylene oxide sterilization, or other recommended techniques for laparoscopic instruments.
Remember to always consult the manufacturer’s instructions and guidelines for the specific sodium perborate monohydrate product you are using, as they may vary. It’s also essential to follow proper safety precautions, including wearing appropriate personal protective equipment, when handling any cleaning or sterilizing agents.
Sodium perborate is a chemical compound that is used as a cleaning and disinfecting agent. It is a white powder that dissolves in water to form hydrogen peroxide and boric acid. These two chemicals work together to kill bacteria, viruses, and fungi.
For laparoscopic instruments, sodium perborate is used in a process called peracetic acid (PAA) decontamination. PAA is a powerful germicide that is effective against a wide range of microorganisms. It is also relatively non-corrosive, making it a good choice for cleaning delicate instruments.
The process of PAA decontamination with sodium perborate begins with the instruments being soaked in a solution of sodium perborate and water. The solution is then heated to a temperature of 130-140 degrees Fahrenheit. This causes the sodium perborate to decompose, releasing hydrogen peroxide and boric acid. The hydrogen peroxide then kills the microorganisms on the instruments.
The PAA decontamination process is typically followed by a rinse with sterile water to remove any residual chemicals. The instruments are then ready to be used again.
Here are some of the advantages of using sodium perborate for laparoscopic instruments:
However, there are also some disadvantages to using sodium perborate:
1. Pre-cleaning: Start by removing any visible debris or organic matter from the instruments. Rinse them with water to remove initial contaminants.
2. Preparation: Prepare a cleaning solution by dissolving sodium perborate monohydrate in water according to the manufacturer’s instructions. The concentration and contact time may vary, so refer to the product label for specific guidelines.
3. Immersion: Submerge the laparoscopic instruments in the prepared cleaning solution, ensuring they are fully covered. Allow them to soak for the recommended duration. This typically ranges from a few minutes to several hours, depending on the concentration and instructions provided by the manufacturer.
4. Agitation: During the soaking period, gently agitate the instruments to facilitate the removal of any remaining debris or contaminants. This can be done by gently swirling the container or using a soft brush to scrub hard-to-reach areas.
5. Rinse: After the recommended contact time, remove the instruments from the cleaning solution and thoroughly rinse them with clean water. Ensure that all residues from the cleaning solution are completely removed.
6. Drying and further sterilization: Once the instruments are rinsed, dry them with a clean, lint-free cloth or allow them to air dry. Keep in mind that sterilization is a separate process, and sodium perborate monohydrate alone may not provide sufficient sterilization. Depending on the specific requirements, you may need to use additional sterilization methods such as autoclaving, ethylene oxide sterilization, or other recommended techniques for laparoscopic instruments.
Remember to always consult the manufacturer’s instructions and guidelines for the specific sodium perborate monohydrate product you are using, as they may vary. It’s also essential to follow proper safety precautions, including wearing appropriate personal protective equipment, when handling any cleaning or sterilizing agents.
Sodium perborate is a chemical compound that is used as a cleaning and disinfecting agent. It is a white powder that dissolves in water to form hydrogen peroxide and boric acid. These two chemicals work together to kill bacteria, viruses, and fungi.
For laparoscopic instruments, sodium perborate is used in a process called peracetic acid (PAA) decontamination. PAA is a powerful germicide that is effective against a wide range of microorganisms. It is also relatively non-corrosive, making it a good choice for cleaning delicate instruments.
The process of PAA decontamination with sodium perborate begins with the instruments being soaked in a solution of sodium perborate and water. The solution is then heated to a temperature of 130-140 degrees Fahrenheit. This causes the sodium perborate to decompose, releasing hydrogen peroxide and boric acid. The hydrogen peroxide then kills the microorganisms on the instruments.
The PAA decontamination process is typically followed by a rinse with sterile water to remove any residual chemicals. The instruments are then ready to be used again.
Here are some of the advantages of using sodium perborate for laparoscopic instruments:
- It is effective against a wide range of microorganisms.
- It is relatively non-corrosive, making it a good choice for cleaning delicate instruments.
- It is easy to use and does not require specialized equipment.
However, there are also some disadvantages to using sodium perborate:
- It can be corrosive to some metals, such as copper and brass.
- It can be irritating to the skin and eyes.
- It is not as effective as some other disinfectants, such as glutaraldehyde.

average cost of retin a